Potassium chlorite
Chemical compound
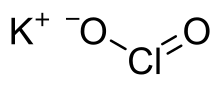 | |
| Names | |
|---|---|
| IUPAC name potassium;chlorite | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| MeSH | 67063160 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | KClO2, ClKO2 |
| Molar mass | 106.55 g/mol |
| Hazards | |
| GHS labelling: | |
Pictograms |  |
Hazard statements | H314 |
Precautionary statements | P260, P264, P280, P301+P330+P331, P304+P340, P305+P351+P338, P310, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Potassium chlorite is a potassium salt of chlorous acid (HClO2) having a chemical formula KClO2. It exists as white powder and its annhydrous form easily undergoes decomposition in presence of heat or radiation (especially gamma rays).[1]
Properties
Potassium chlorite is a colorless hygroscopic crystal that deliquesces in the air. It decomposes upon heating into potassium chloride and oxygen, emitting light.
Potassium chlorite forms orthorhombic cmcm crystals and has been reported to decompose within hours at room temperature.[1][2] It is an oxidizing agent.
Synthesis
Some of the methods of preparation of potassium chlorite are:
- Thermal decomposition of potassium chlorate
- Reaction of chloric acid and potassium hydroxide
References
- ^ a b Boyd, George E.; Brown, Larry Clyde (1970). "Thermal and radiolytic decomposition of anhydrous crystalline potassium chlorite". The Journal of Physical Chemistry. 74 (8): 1691–1694. doi:10.1021/j100703a006. ISSN 0022-3654.
- ^ Smolentsev, A. I.; Naumov, D. Yu (2005-02-15). "Two alkali metal chlorites, LiClO2 and KClO2". Acta Crystallographica Section C: Crystal Structure Communications. 61 (2): i17–i19. doi:10.1107/S0108270104032482. ISSN 0108-2701.
- v
- t
- e
- K3N
- KNH2
- KN3
- KNO2
- KNO3
- K3P
- KH2PO3
- K3PO4
- K2HPO4
- KH2PO4
- KPF6
- KAsO2
- K3AsO4
- K2HAsO4
- KH2AsO4
- B4K2O7
- K2CO3
- KHCO3
- K2SiO3
- K2SiF6
- K2Al2O4
- K2Al2B2O7
- K2PtCl4
- K2Pt(CN)4
- K2TiF6
- K2PtCl6
- K2ReCl6
- K
2ReF
6 - KAsF6
- K2ReBr6
- K
2ReI
6 - K2ZrF6
- K4Fe(CN)6
- K3Fe(CN)6
- K3Fe(C2O4)3
- K2FeO4
- K2MnO4
- KMnO4
- K3CrO4
- K2CrO4
- K3CrO8
- KCrO3Cl
- K2Cr2O7
- K2Cr3O10
- K2Cr4O13
- K4Mo2Cl8
- KHCO2
- KCH3CO2
- KCF3CO2
- K2C2O4
- KHC2O4
- KC12H23O2
- KC18H35O2
- C3H2K2O4
- C4H6KO4
- C5H7KO4
 | This article about chemical compounds is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e














